France Medical Composites Market (2025-2031) Outlook | Forecast, Value, Revenue, Industry, Trends, Growth, Analysis, Companies, Share, Size
| Product Code: ETC4499531 | Publication Date: Jul 2023 | Updated Date: Nov 2025 | Product Type: Report | |
| Publisher: 6Wresearch | Author: Ravi Bhandari | No. of Pages: 85 | No. of Figures: 45 | No. of Tables: 25 |
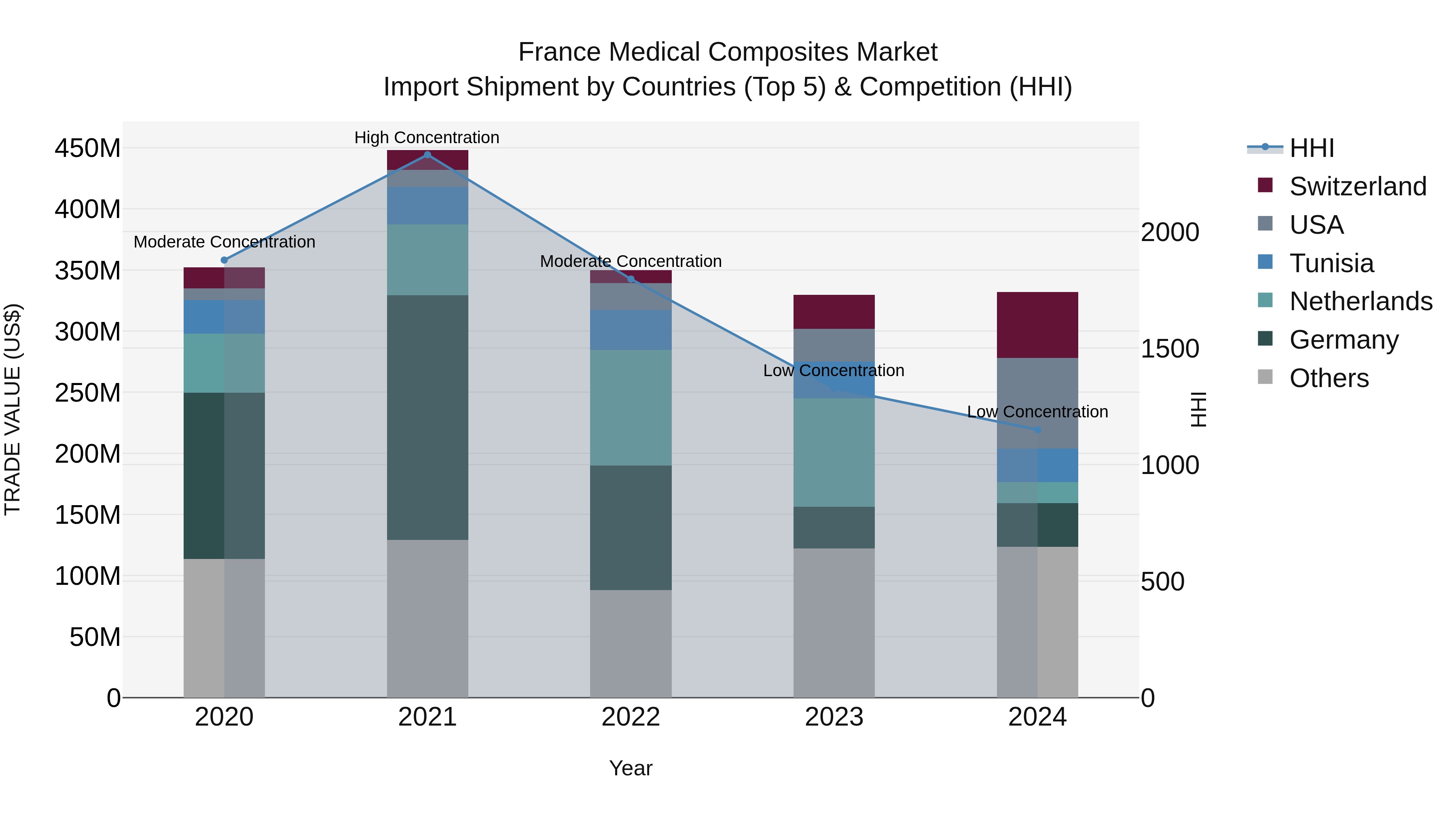
France Medical Composites Market Top 5 Importing Countries and Market Competition (HHI) Analysis
Despite a slight decline in the compound annual growth rate from 2020 to 2024, the medical composites import market in France saw a positive growth rate in 2024. The top exporting countries to France, including the USA, Switzerland, Germany, Poland, and Tunisia, indicate a diverse market for medical composites. With a low concentration level measured by the Herfindahl-Hirschman Index (HHI), the market remains competitive and open to various suppliers. The marginal growth rate from 2023 to 2024 suggests a stable trajectory for the import of medical composites in France.
France Medical Composites Market Overview
France medical composites market offers lightweight and biocompatible materials for medical device manufacturing, prosthetics, and orthopedic implants. Medical composites combine polymers, ceramics, and fibers to create strong and durable components for medical applications. With a focus on innovation and regulatory compliance, France medical composites suppliers develop advanced materials to meet the stringent requirements of the healthcare industry.
Drivers of the market
In France, the Medical Composites Market is influenced by factors such as the growing demand for lightweight and high-strength materials in medical devices, the advancements in composite manufacturing technologies, and the increasing applications of composites in healthcare. Medical composites, composed of fibers such as carbon, glass, or aramid embedded in a polymer matrix, offer advantages such as biocompatibility, radiolucency, and corrosion resistance, making them ideal for medical applications such as orthopedic implants, prosthetics, and diagnostic equipment. With medical device manufacturers seeking to innovate in product design, improve patient outcomes, and comply with regulatory standards, there`s a rising demand for medical composites in the French market for medical device manufacturing and healthcare infrastructure.
Challenges of the market
In the France medical composites market, challenges include ensuring biocompatibility, sterilizability, and regulatory compliance for medical devices and implants. Medical composites offer advantages such as lightweight construction, radiolucency, and corrosion resistance, but their implementation requires overcoming technical challenges such as material compatibility, dimensional stability, and long-term durability. Moreover, ensuring traceability in supply chain operations, addressing concerns about implant rejection, and navigating regulatory pathways for product approvals pose ongoing challenges for manufacturers and users in this market.
Government Policy of the market
Government regulations in France govern the use of medical composites in healthcare applications, including standards for material safety, biocompatibility, and product labeling. Regulatory bodies ensure compliance with medical device regulations, quality standards, and patient safety requirements. Additionally, reimbursement policies may influence market adoption by healthcare providers and insurers.
Key Highlights of the Report:
- France Medical Composites Market Outlook
- Market Size of France Medical Composites Market, 2024
- Forecast of France Medical Composites Market, 2031
- Historical Data and Forecast of France Medical Composites Revenues & Volume for the Period 2021-2031
- France Medical Composites Market Trend Evolution
- France Medical Composites Market Drivers and Challenges
- France Medical Composites Price Trends
- France Medical Composites Porter's Five Forces
- France Medical Composites Industry Life Cycle
- Historical Data and Forecast of France Medical Composites Market Revenues & Volume By Fiber Type for the Period 2021-2031
- Historical Data and Forecast of France Medical Composites Market Revenues & Volume By Carbon for the Period 2021-2031
- Historical Data and Forecast of France Medical Composites Market Revenues & Volume By Ceramic for the Period 2021-2031
- Historical Data and Forecast of France Medical Composites Market Revenues & Volume By Application for the Period 2021-2031
- Historical Data and Forecast of France Medical Composites Market Revenues & Volume By Diagnostic Imaging for the Period 2021-2031
- Historical Data and Forecast of France Medical Composites Market Revenues & Volume By Composite Body Implants for the Period 2021-2031
- Historical Data and Forecast of France Medical Composites Market Revenues & Volume By Surgical Instruments for the Period 2021-2031
- Historical Data and Forecast of France Medical Composites Market Revenues & Volume By Dental for the Period 2021-2031
- France Medical Composites Import Export Trade Statistics
- Market Opportunity Assessment By Fiber Type
- Market Opportunity Assessment By Application
- France Medical Composites Top Companies Market Share
- France Medical Composites Competitive Benchmarking By Technical and Operational Parameters
- France Medical Composites Company Profiles
- France Medical Composites Key Strategic Recommendations
Frequently Asked Questions About the Market Study (FAQs):
1 Executive Summary |
2 Introduction |
2.1 Key Highlights of the Report |
2.2 Report Description |
2.3 Market Scope & Segmentation |
2.4 Research Methodology |
2.5 Assumptions |
3 France Medical Composites Market Overview |
3.1 France Country Macro Economic Indicators |
3.2 France Medical Composites Market Revenues & Volume, 2021 & 2031F |
3.3 France Medical Composites Market - Industry Life Cycle |
3.4 France Medical Composites Market - Porter's Five Forces |
3.5 France Medical Composites Market Revenues & Volume Share, By Fiber Type, 2021 & 2031F |
3.6 France Medical Composites Market Revenues & Volume Share, By Application, 2021 & 2031F |
4 France Medical Composites Market Dynamics |
4.1 Impact Analysis |
4.2 Market Drivers |
4.2.1 Increasing demand for lightweight and high-strength materials in the healthcare sector |
4.2.2 Rising adoption of medical composites in orthopedic and dental applications |
4.2.3 Technological advancements leading to the development of innovative medical composite materials |
4.3 Market Restraints |
4.3.1 High manufacturing costs associated with medical composites |
4.3.2 Stringent regulatory requirements for medical devices using composites |
4.3.3 Limited awareness and adoption of medical composites in certain healthcare segments |
5 France Medical Composites Market Trends |
6 France Medical Composites Market, By Types |
6.1 France Medical Composites Market, By Fiber Type |
6.1.1 Overview and Analysis |
6.1.2 France Medical Composites Market Revenues & Volume, By Fiber Type, 2021-2031F |
6.1.3 France Medical Composites Market Revenues & Volume, By Carbon , 2021-2031F |
6.1.4 France Medical Composites Market Revenues & Volume, By Ceramic, 2021-2031F |
6.2 France Medical Composites Market, By Application |
6.2.1 Overview and Analysis |
6.2.2 France Medical Composites Market Revenues & Volume, By Diagnostic Imaging, 2021-2031F |
6.2.3 France Medical Composites Market Revenues & Volume, By Composite Body Implants, 2021-2031F |
6.2.4 France Medical Composites Market Revenues & Volume, By Surgical Instruments, 2021-2031F |
6.2.5 France Medical Composites Market Revenues & Volume, By Dental, 2021-2031F |
7 France Medical Composites Market Import-Export Trade Statistics |
7.1 France Medical Composites Market Export to Major Countries |
7.2 France Medical Composites Market Imports from Major Countries |
8 France Medical Composites Market Key Performance Indicators |
8.1 Research and development investment in medical composite materials |
8.2 Number of patents filed for new medical composite technologies |
8.3 Adoption rate of medical composites in new medical device applications |
9 France Medical Composites Market - Opportunity Assessment |
9.1 France Medical Composites Market Opportunity Assessment, By Fiber Type, 2021 & 2031F |
9.2 France Medical Composites Market Opportunity Assessment, By Application, 2021 & 2031F |
10 France Medical Composites Market - Competitive Landscape |
10.1 France Medical Composites Market Revenue Share, By Companies, 2024 |
10.2 France Medical Composites Market Competitive Benchmarking, By Operating and Technical Parameters |
11 Company Profiles |
12 Recommendations |
13 Disclaimer |
Export potential assessment - trade Analytics for 2030
Export potential enables firms to identify high-growth global markets with greater confidence by combining advanced trade intelligence with a structured quantitative methodology. The framework analyzes emerging demand trends and country-level import patterns while integrating macroeconomic and trade datasets such as GDP and population forecasts, bilateral import–export flows, tariff structures, elasticity differentials between developed and developing economies, geographic distance, and import demand projections. Using weighted trade values from 2020–2024 as the base period to project country-to-country export potential for 2030, these inputs are operationalized through calculated drivers such as gravity model parameters, tariff impact factors, and projected GDP per-capita growth. Through an analysis of hidden potentials, demand hotspots, and market conditions that are most favorable to success, this method enables firms to focus on target countries, maximize returns, and global expansion with data, backed by accuracy.
By factoring in the projected importer demand gap that is currently unmet and could be potential opportunity, it identifies the potential for the Exporter (Country) among 190 countries, against the general trade analysis, which identifies the biggest importer or exporter.
To discover high-growth global markets and optimize your business strategy:
Click Here- Single User License$ 1,995
- Department License$ 2,400
- Site License$ 3,120
- Global License$ 3,795
Search
Thought Leadership and Analyst Meet
Our Clients
Latest Reports
- India Ultrasonic Smart Water Meter Market (2026-2032) | Outlook, Competition, Drivers, Trends, Demand, Competitive, Strategic Insights, Companies, Challenges, Strategy, Consumer Insights, Analysis, Opportunities, Growth, Size, Share, Industry, Revenue, Segments, Value, Segmentation, Supply, Forecast, Restraints,
- India Electromagnetic Bulk Flow Meter Market (2026-2032) | Analysis, Challenges, Strategic Insights, Supply, Forecast, Growth, Share, Consumer Insights, Competitive, Value, Outlook, Size & Demand,
- India Battery Powered Bulk Flow Meter Market (2026-2032) | Outlook, Analysis, Competitive Landscape, Growth, Size, Trends, Value, Revenue, Companies, Segmentation, Share & Forecast
- Bosnia and Herzegovina Fuel-Efficient Winglets Market (2026-2032) | Growth, Competition, Strategy, Restraints, Trends, Challenges, segmentation, Value, Outlook, Investment Opportunities, Analysis, Revenue, Companies, Insights, Pricing, Size, Drivers, Demand, Share, Forecast
- Canada Cloud CFD Market (2026-2032) | Size & Revenue, Industry, Growth, Competitive Landscape, Forecast, Segmentation, Value, Outlook, Trends, Share, Analysis, Companies
- Taiwan Food Delivery Platform Market (2026-2032) | Companies, Outlook, Analysis, Trends, Value, Revenue, Segmentation, Share, Forecast, Competitive Landscape, Growth, Size & Forecast
- United Kingdom (UK) Long-term Care Insurance Market (2026-2032) | Growth, Share, Consumer Insights, Drivers, Opportunities, Competition, Pricing Analysis, Segments, Restraints, Companies, Competitive, Value, Outlook, Size, Demand, Analysis, Challenges, Strategic Insights, Investment Trends, Revenue, Trends, Supply, Forecast
- United Kingdom (UK) Long Term Care Market (2026-2032) | Companies, Outlook, Analysis, Trends, Value, Revenue, Segmentation, Share, Forecast, Competitive Landscape, Growth, Size & Forecast
- Iraq Insulation and Waterproofing Market (2026-2032) | Outlook, Drivers, Growth, Size, Share, Industry, Revenue, Trends, Demand, Competitive, Strategic Insights, Opportunities, Segments, Companies, Challenges, Strategy, Consumer Insights, Analysis, Investment Trends, Value, Segmentation, Forecast, Restraints
- India Switchgear Market Outlook (2026-2032) | Size, Share, Trends, Growth, Revenue, Forecast, Analysis, Value, Outlook
Industry Events and Analyst Meet
Whitepaper
- Middle East & Africa Commercial Security Market Click here to view more.
- Middle East & Africa Fire Safety Systems & Equipment Market Click here to view more.
- GCC Drone Market Click here to view more.
- Middle East Lighting Fixture Market Click here to view more.
- GCC Physical & Perimeter Security Market Click here to view more.
6WResearch In News
- Doha a strategic location for EV manufacturing hub: IPA Qatar
- Demand for luxury TVs surging in the GCC, says Samsung
- Empowering Growth: The Thriving Journey of Bangladesh’s Cable Industry
- Demand for luxury TVs surging in the GCC, says Samsung
- Video call with a traditional healer? Once unthinkable, it’s now common in South Africa
- Intelligent Buildings To Smooth GCC’s Path To Net Zero


















